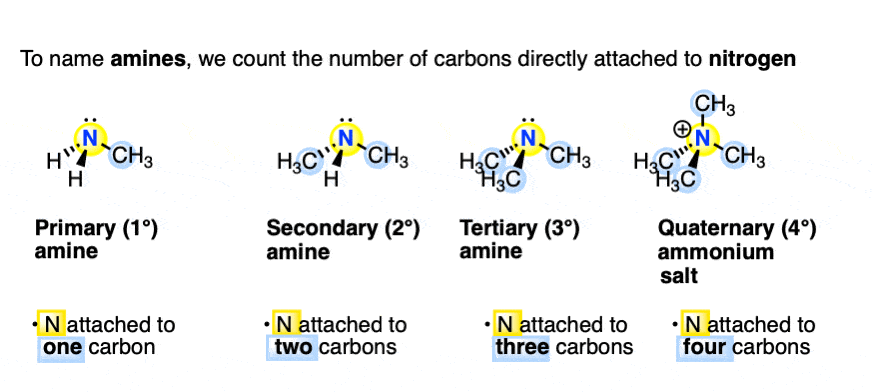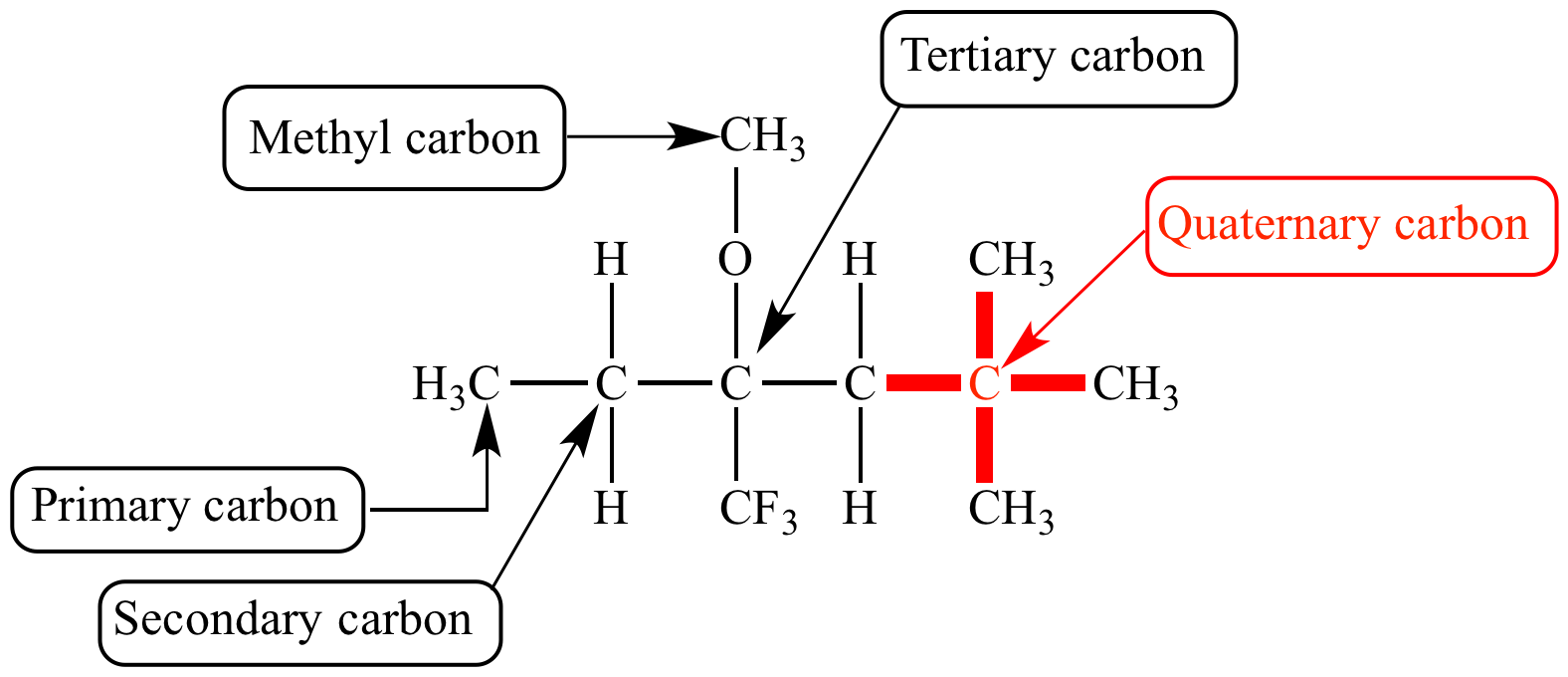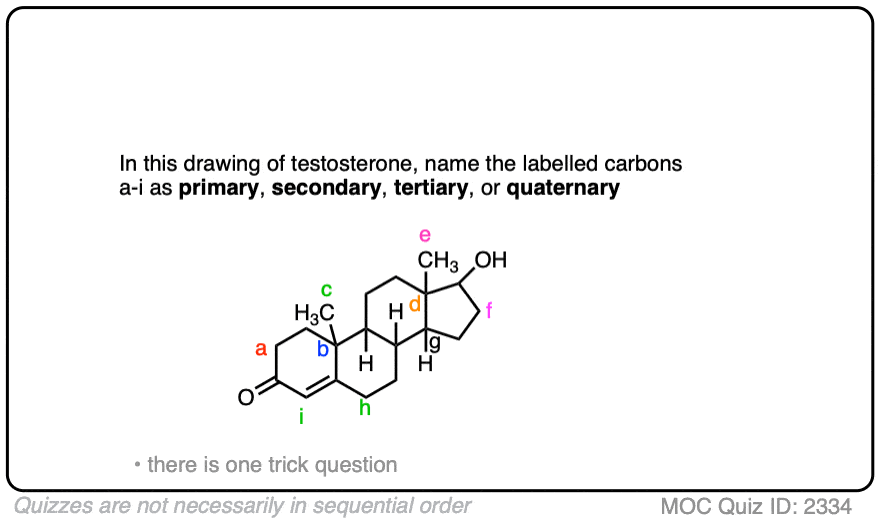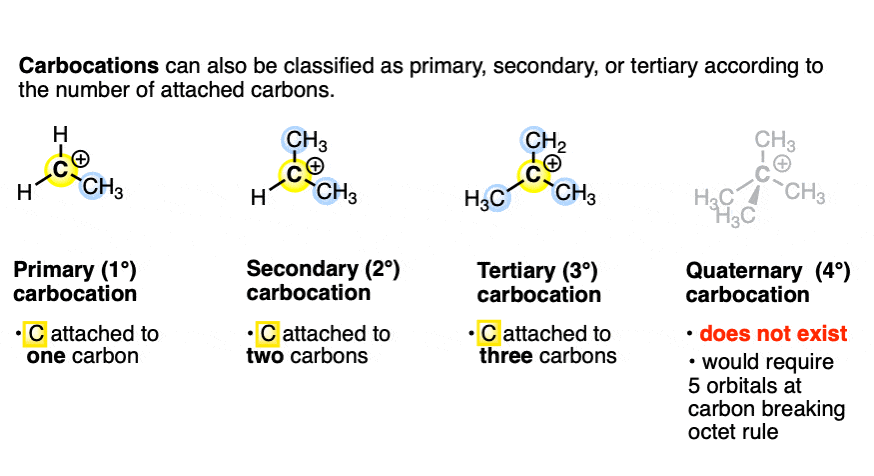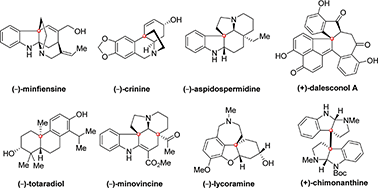
Enantioselective formation of quaternary carbon stereocenters in natural product synthesis: a recent update - Natural Product Reports (RSC Publishing)

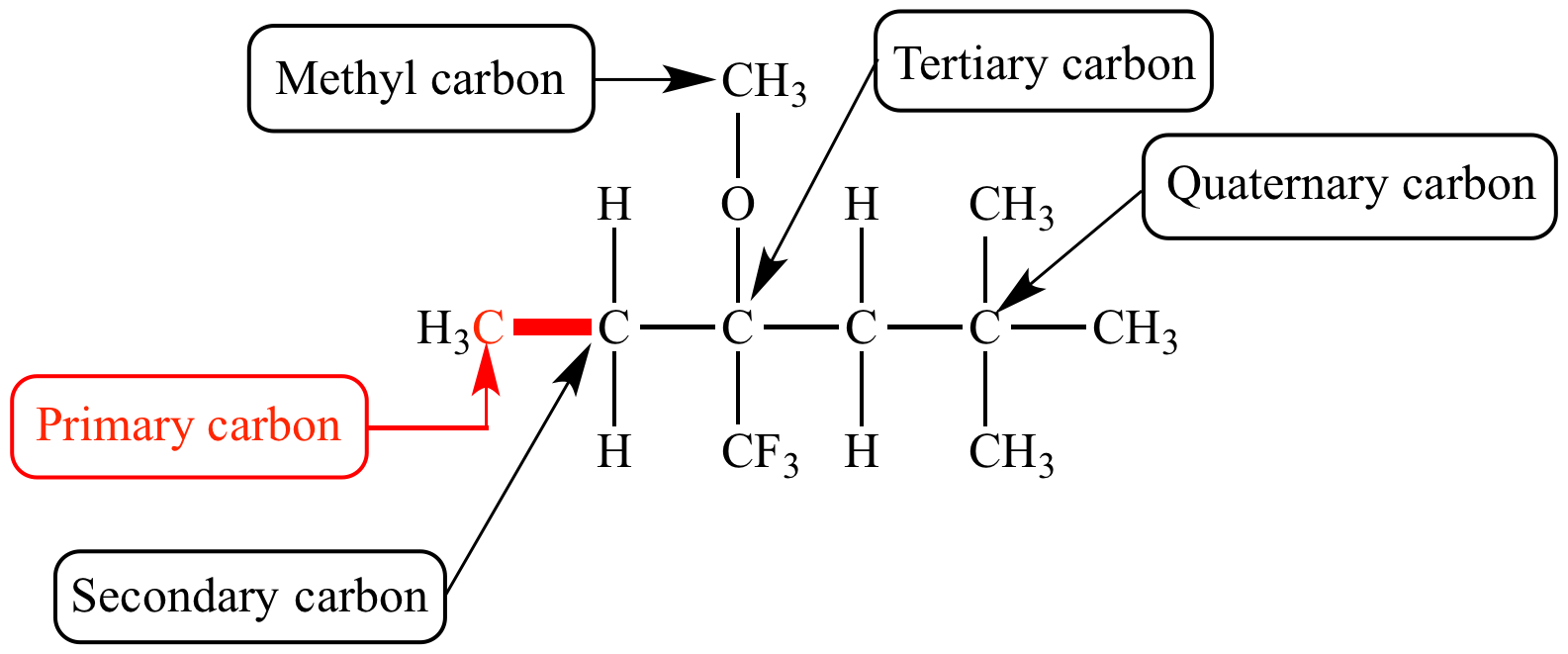
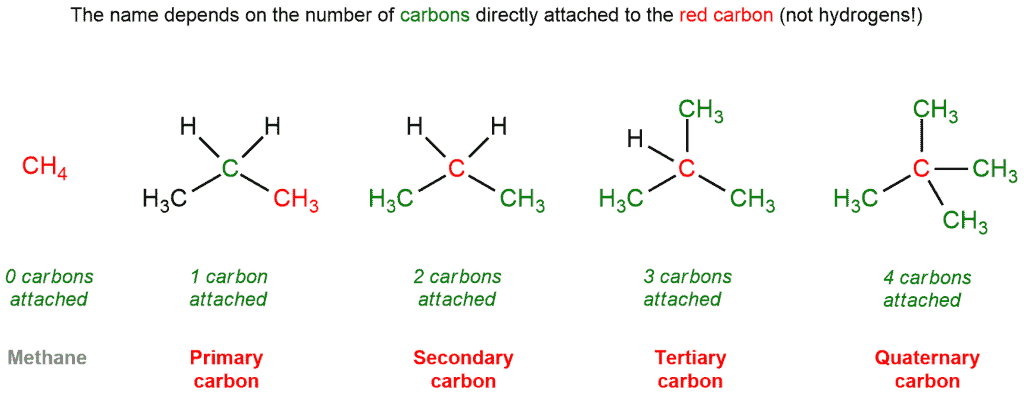
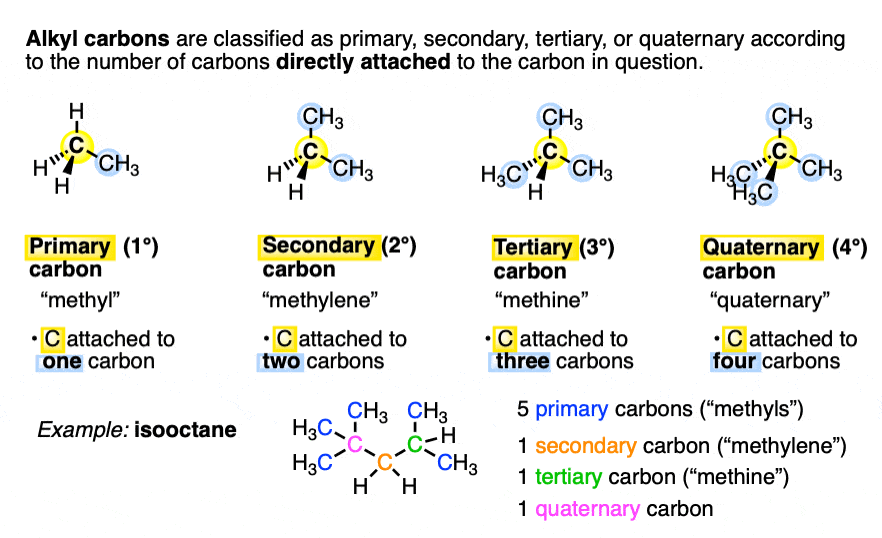
Identify primary, secondary, tertiary, and quaternary carbon in the following compound. CHX3..................|....................CHX3−C−CH−CHX2−CHX2−CHX3|.....|................CHX3.CHX3............. - Chemistry | Shaalaa.com

Enantioselective Synthesis of α-All-Carbon Quaternary Center-Containing Carbazolones via Amino-palladation/Desymmetrizing Nitrile Addition Cascade | Journal of the American Chemical Society

Construction of Acyclic Quaternary Carbon Stereocenters by Catalytic Asymmetric Hydroalkynylation of Unactivated Alkenes | Journal of the American Chemical Society
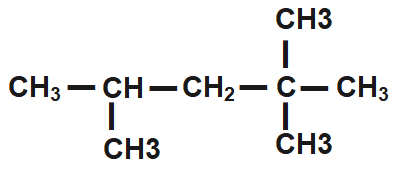
How many primary, secondary, tertiary, and quaternary carbon atoms are present in the following compound?\n \n \n \n \n (A) One primary, two secondary and one tertiary.(B) Five Primary, three secondary.(C) Five
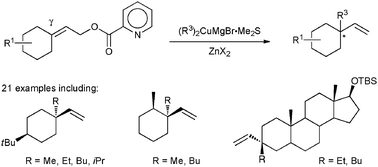
Construction of a quaternary carbon at the carbonyl carbon of the cyclohexane ring - Chemical Communications (RSC Publishing)

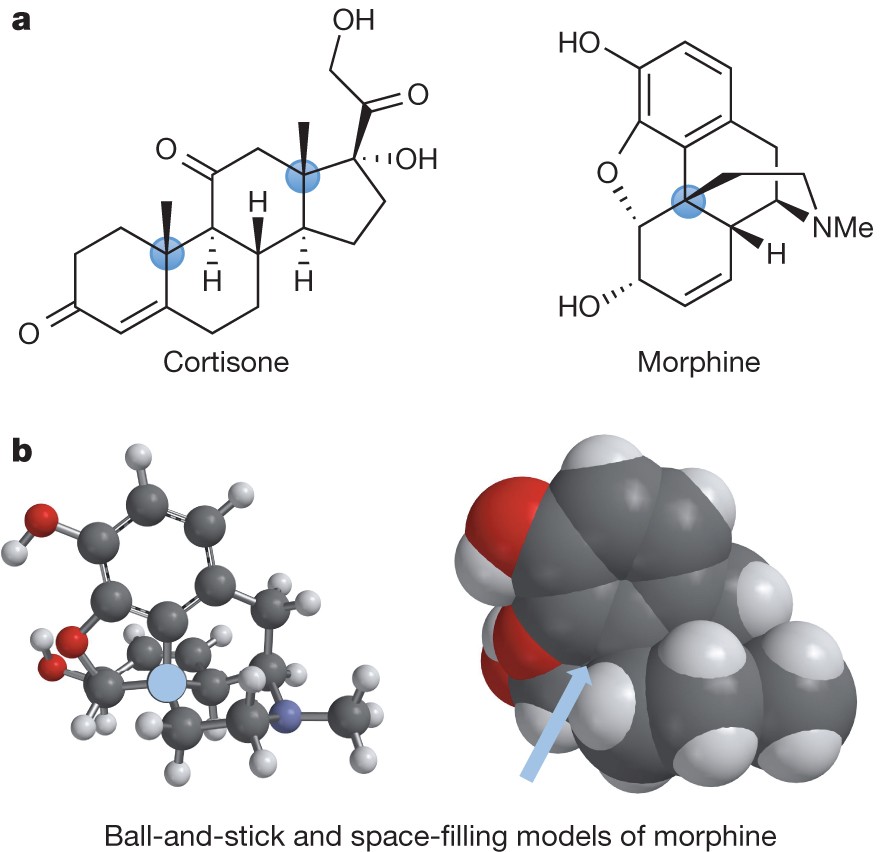
Quaternary centres as a point of challenge and opportunity for organic... | Download Scientific Diagram

CH3 - CH3 |CH - CH2 - CH3 | CH3 |C - CH3 How many primary, secondary, tertiary and quaternary carbon atoms are present in the following compound?

Synthesis of Quaternary Carbon-Centered Benzoindolizidinones via Novel Photoredox-Catalyzed Alkene Aminoarylation: Facile Access to Tylophorine and Analogues | CCS Chem