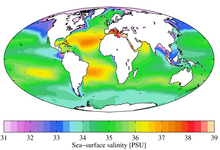
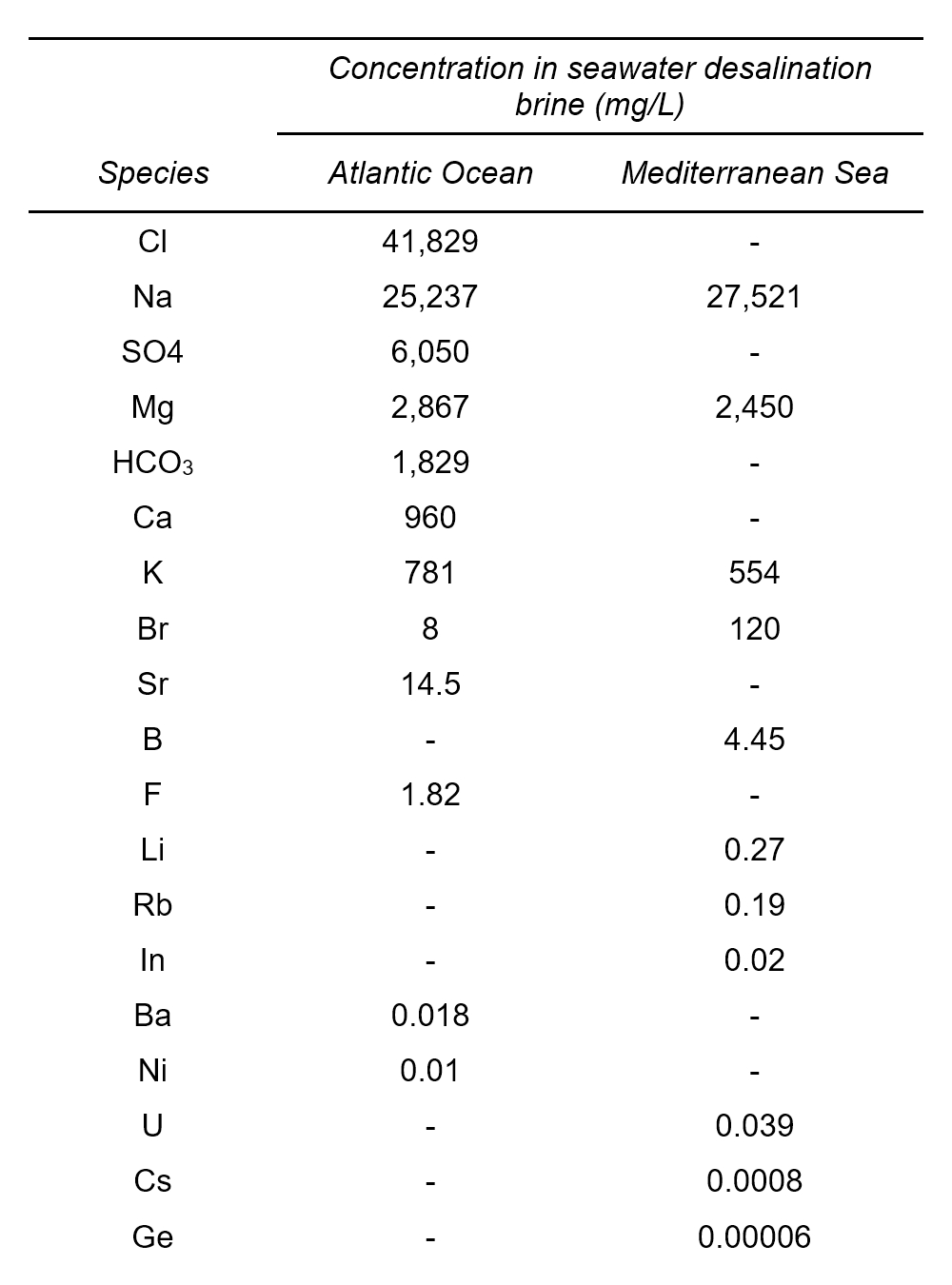
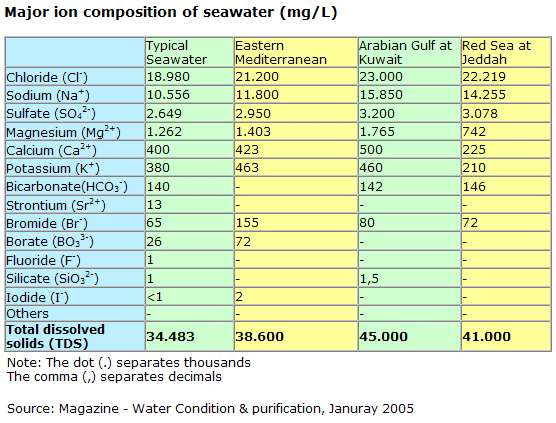
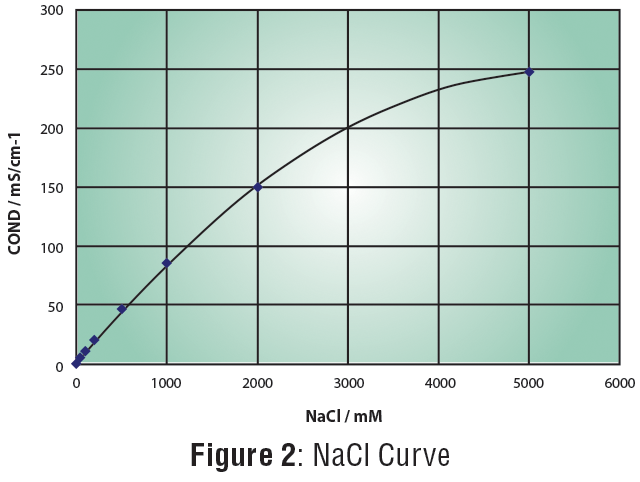
Concentration of NaCl from seawater reverse osmosis brines for the chlor-alkali industry by electrodialysis - ScienceDirect
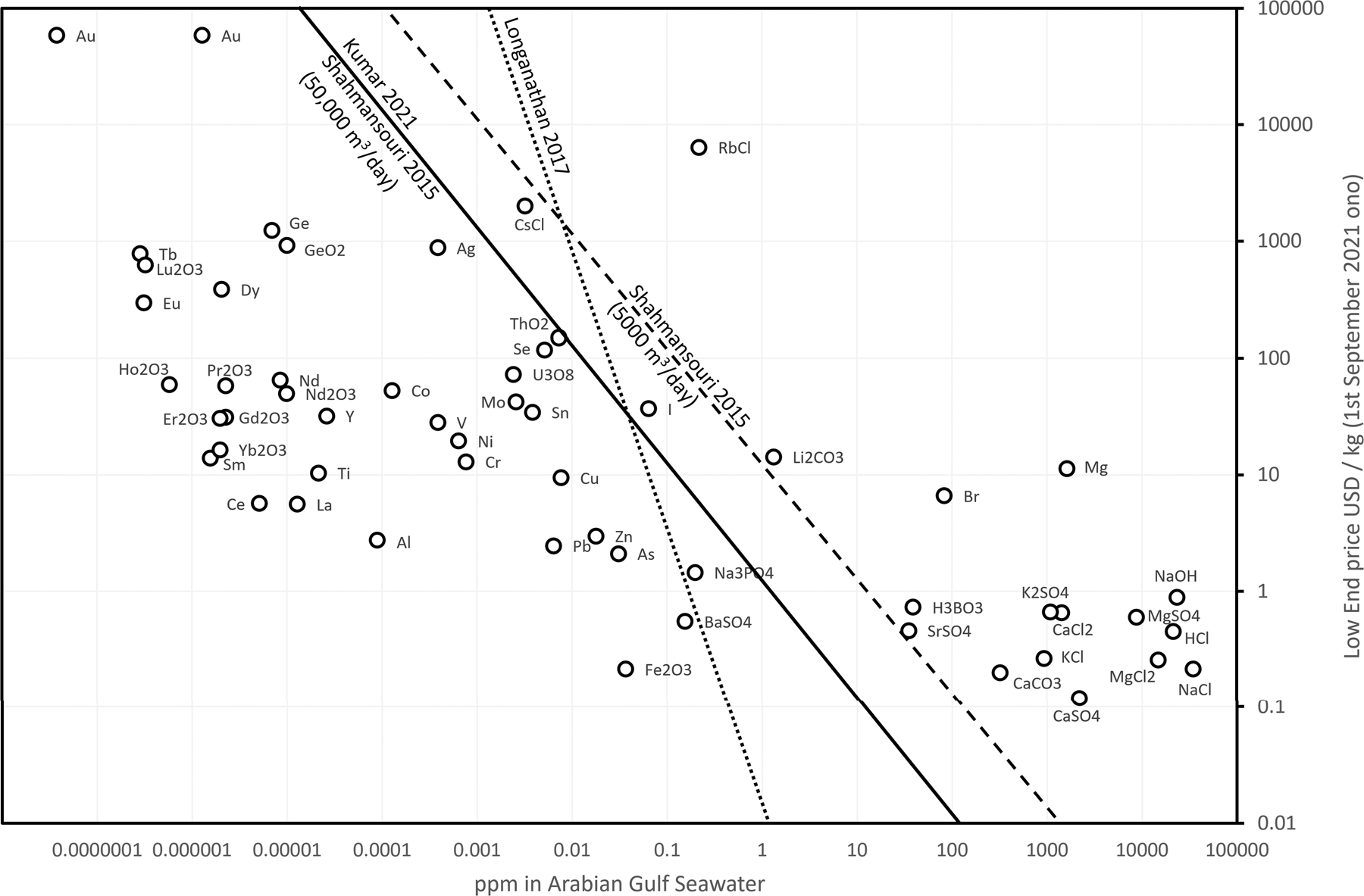
Seawater desalination concentrate—a new frontier for sustainable mining of valuable minerals | npj Clean Water
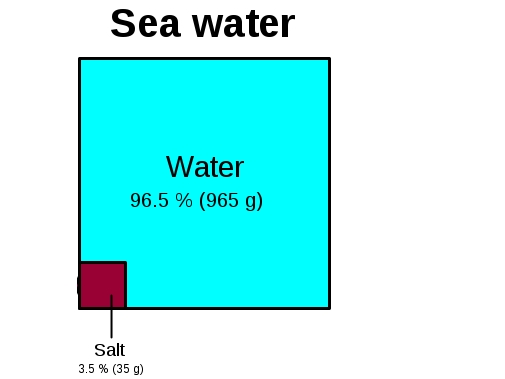
Assuming that sea water is an aqueous solution of NaCl its density is 1.025 g/mL at 20°C and NaCl concentration is 3.5% - Sarthaks eConnect | Largest Online Education Community

Concentration of salts in prepared seawater, based on concentrations... | Download Scientific Diagram

SOLVED:Assuming that seawater is an aqueous solution of NaCl , what is its molarity? The density of seawater is 1.025 g / mL at 20^∘ C, and the NaCl concentration is 3.50 mass % .
![SOLVED: Seawater is approximately 0.438M NaCl and 0.0512M MgCl2. What is the molarity of Cl^-(that is the total [Cl^-] in seawater? SOLVED: Seawater is approximately 0.438M NaCl and 0.0512M MgCl2. What is the molarity of Cl^-(that is the total [Cl^-] in seawater?](https://cdn.numerade.com/ask_previews/1b631a19-e659-4707-8efa-2d3b24e45458_large.jpg)