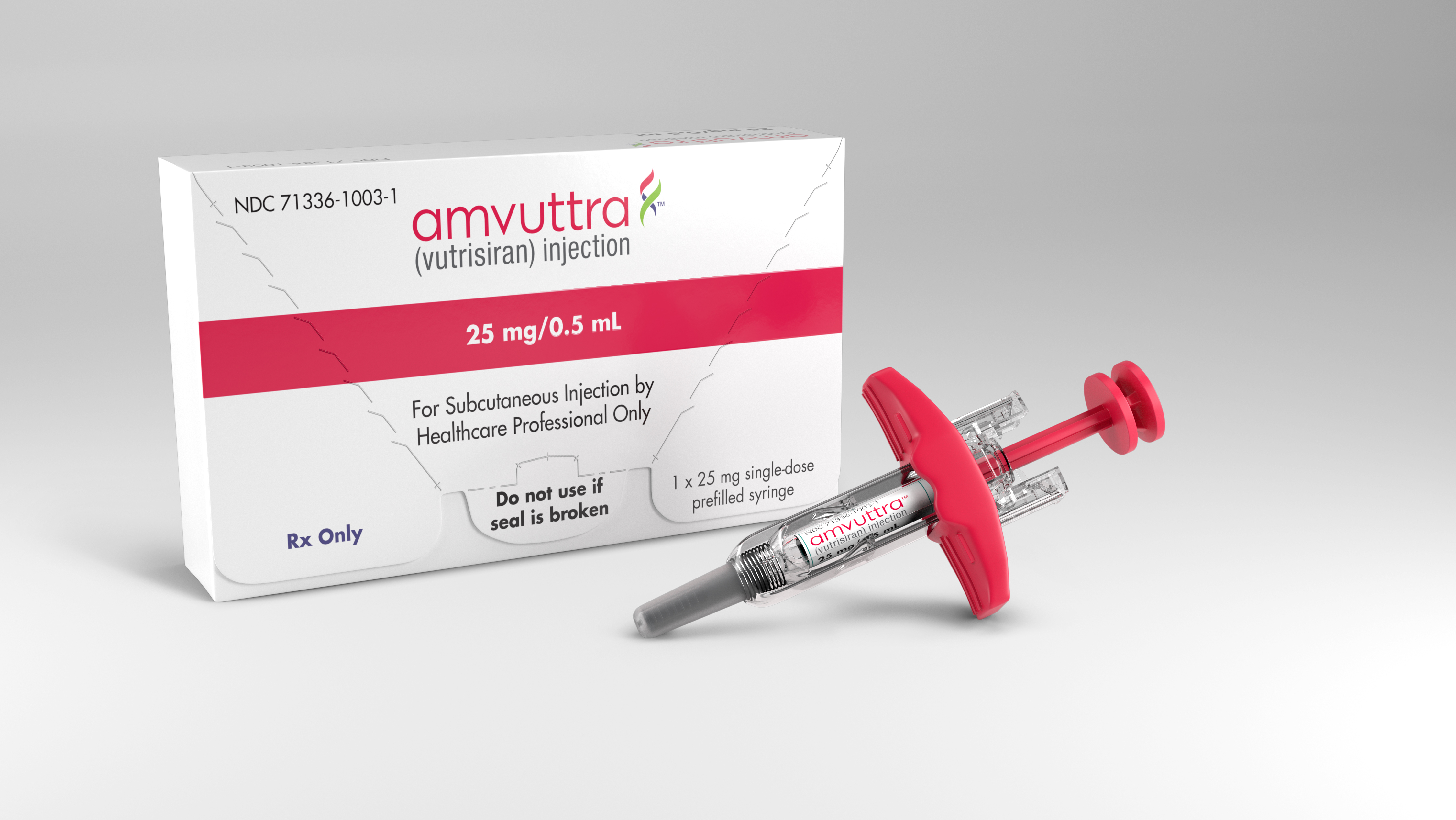
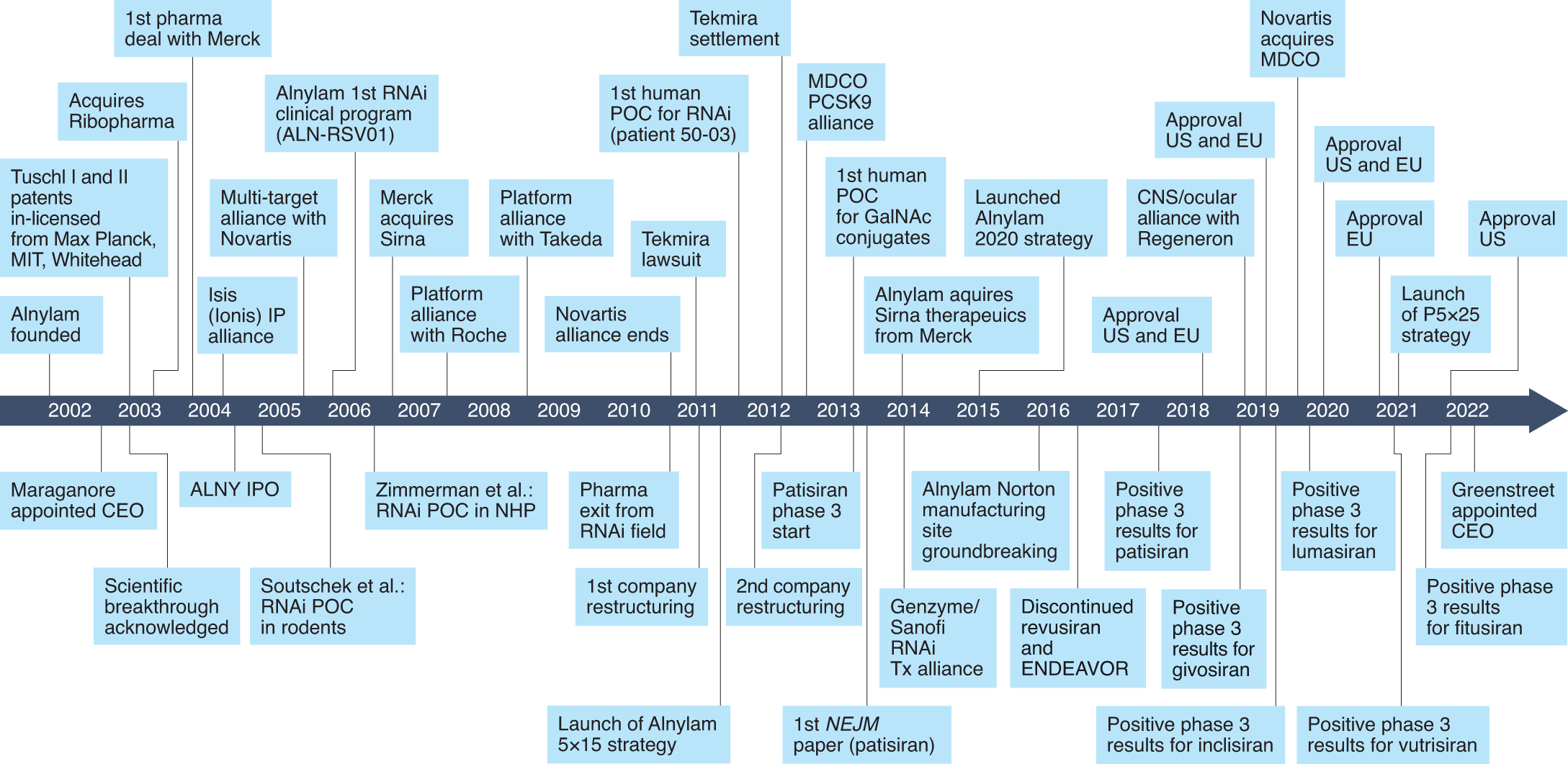
Alnylam Announces First-Ever FDA Approval of an RNAi Therapeutic, ONPATTRO™ (patisiran) for the Treatment of the Polyneuropathy of Hereditary Transthyretin-Mediated Amyloidosis in Adults | Business Wire

Dr. Yvonne Greenstreet on LinkedIn: Alnylam Announces Preliminary* Fourth Quarter and Full Year 2022 Global…

Novartis anuncia colaboración con Alnylam Pharmaceuticals para explorar terapia para restaurar función hepática - enFarma

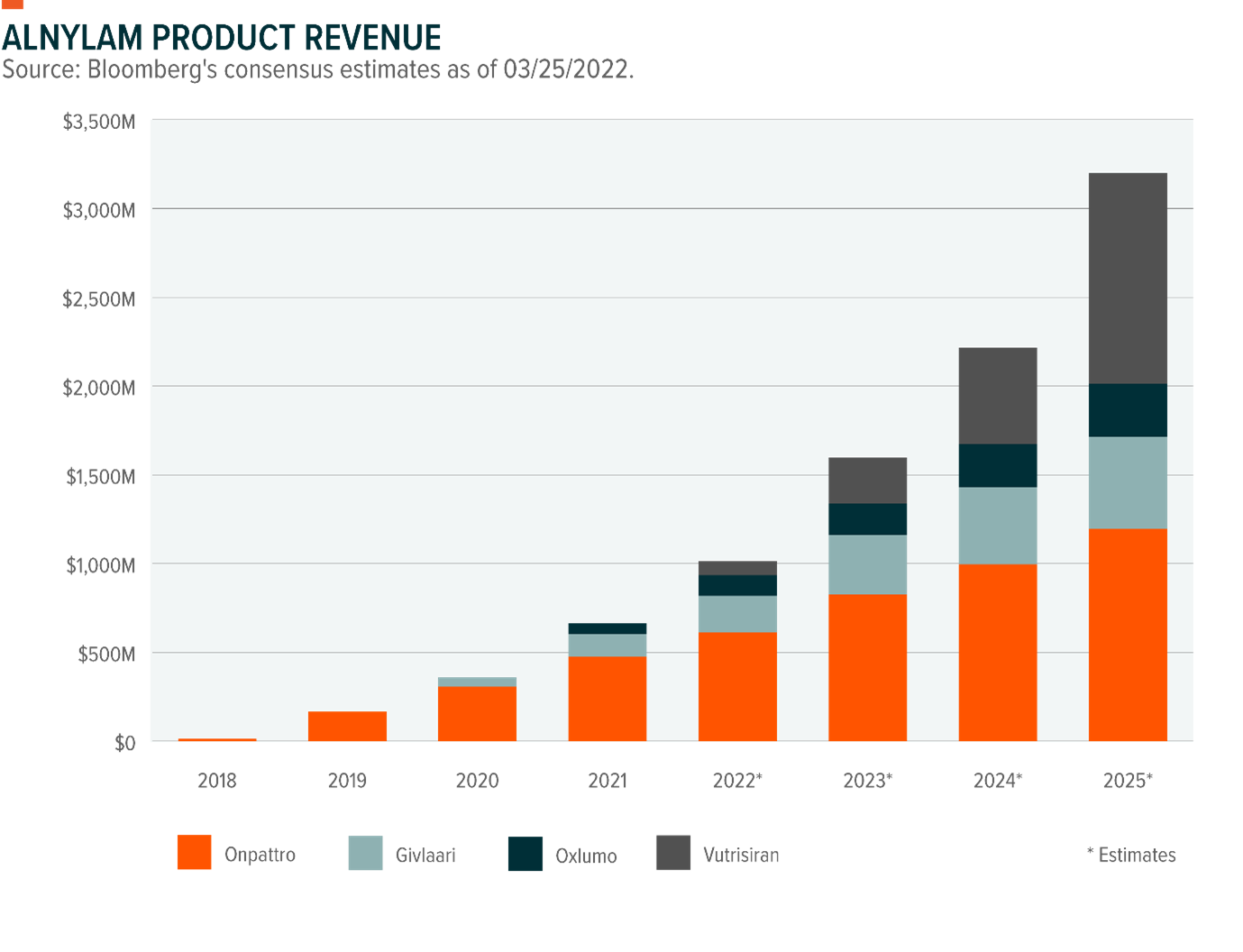
Alnylam Announces 2023 Product and Pipeline Goals and Provides Program Updates at R&D Day | Business Wire
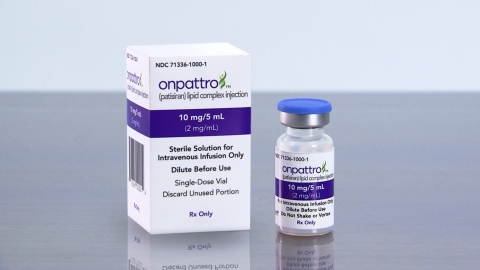
Alnylam Pharmaceuticals Press Release | Aug 10, 2018 | Alnylam Announces First-Ever FDA Approval of an RNAi Therapeutic, ONPATTRO™ (patisiran) for the Treatment of

Alnylam Pharmaceuticals Press Release | Nov 24, 2020 | Alnylam Announces U.S. Food and Drug Administration (FDA) Approval of OXLUMO™ (lumasiran), the First and Onl